PSORIASIS
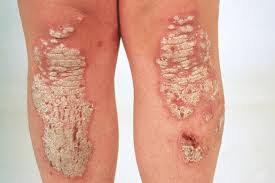
Psoriasis is a skin disorder that causes skin cells to multiply up to 10 times faster than normal. This makes the skin build up into bumpy red patches covered with white scales. They can grow anywhere, but most appear on the scalp, elbows, knees, and lower back. Psoriasis can't be passed from person to person. It does sometimes happen in members of the same family.
 PSORIASIS
PSORIASIS
Psoriasis usually appears in early adulthood. For most people, it affects just a few areas. In severe cases, psoriasis can cover large parts of the body. The patches can heal and then come back throughout a person's life.get the basics on psoriasis
SYMPTOMS
The symptoms of psoriasis vary depending on the type you have. Some common symptoms for plaque psoriasis -- the most common variety of the condition -- include
Plaques of red skin, often covered with silver-colored scales. These plaques may be itchy and painful, and they sometimes crack and bleed. In severe cases, the plaques will grow and merge, covering large areas.
Disorders of the fingernails and toenails, including discoloration and pitting of the nails. The nails may also crumble or detach from the nail bed.
Plaques of scales or crust on the scalp.
People with psoriasis can also get a type of arthritis called psoriatic arthritis. It causes pain and swelling in the joints. The National Psoriasis Foundation estimates that between 10% to 30% of people with psoriasis also have psoriatic arthritis.
TYPES
Other types of psoriasis include:
PUSTULAR PSORIASIS- which causes red and scaly skin with tiny pustules on the palms of the hands and soles of the feet.
GUTTATE PSORIASIS- which often starts in childhood or young adulthood, causes small, red spots, mainly on the torso and limbs. Triggers may be respiratory infections, strep throat, tonsillitis, stress, injury to the skin, and taking antimalarial and beta-blocker medications.
INVERSE PSORIASIS- which makes bright red, shiny lesions that appear in skin folds, such as the armpits, groin, and under the breasts.
ERYTHRODERMIC PSORIASIS- which causes fiery redness of the skin and shedding of scales in sheets. It's triggered by severe sunburn, infections, certain medications, and stopping some kinds of psoriasis treatment. It needs to be treated immediately because it can lead to severe illness.
CAUSES
No one knows the exact cause of psoriasis, but experts believe that it’s a combination of things. Something wrong with the immune system causes inflammation, triggering new skin cells to form too quickly. Normally, skin cells are replaced every 10 to 30 days. With psoriasis, new cells grow every 3 to 4 days. The buildup of old cells being replaced by new ones creates those silver scales.
Psoriasis tends to run in families, but it may be skip generations. For instance, a grandfather and his grandson may be affected, but not the child's mother.
Things that can trigger an outbreak of psoriasis include:
- Cuts, scrapes, or surgery
- Emotional stress
- Strep infections
- Medications, including
- Blood pressure medications (like beta-blockers)
- Hydroxychloroquine, antimalarial medication
- Diagnosis
- Physical exam. It’s usually easy for your doctor to diagnose psoriasis, especially if you have plaques on areas such as your:
- Scalp
- Ears
- Elbows
- Knees
- Belly button
- Nails
Lab tests. The doctor might do a biopsy -- remove a small piece of skin and test it to make sure you don’t have a skin infection. There’s no other test to confirm or rule out psoriasis.
TREATMENT
Luckily, there are many treatments. Some slow the growth of new skin cells, and others relieve itching and dry skin. Your doctor will select a treatment plan that is right for you based on the size of your rash, where it is on your body, your age, your overall health, and other things. Common treatments include:
STEROIDS CREAM
Moisturizers for dry skin
Coal tar (a common treatment for scalp psoriasis available in lotions, creams, foams, shampoos, and bath solutions)
cream or ointment (a strong kind ordered by your doctor. Vitamin D in foods and pills has no effect.)
Retinoid creams
Treatments for moderate to severe psoriasis include:
LIGHT THERAPY- A doctor shines ultraviolet light on your skin to slow the growth of skin cells. PUVA is a treatment that combines a medicine called psoralen with a special form of ultraviolet light.
 PSORIASIS LIGHT THERAPY
METHOTRAXATE
PSORIASIS LIGHT THERAPY
METHOTRAXATE- This drug can cause bone marrow and liver disease as well as lung problems, so it’s only for serious cases. Doctors closely watch patients. You will have to get lab tests, perhaps a chest X-ray, and possibly a liver biopsy.
RETINOIDS These pills, creams, foams, lotions, and gels are a class of drugs related to vitamin A. Retinoids can cause serious side effects, including birth defects, so they’re not recommended for women who are pregnant or planning to have children.
. This drug, made to suppress the immune system, may be taken for serious cases that do not respond to other treatments. It can damage the kidneys and raise blood pressure, so your doctor will closely watch your health while you take it.
BIOLOGIC TREATMENT-These work by blocking the body's immune system (which is overactive in psoriasis) to better control the inflammation from psoriasis. Biologic medications include adalimumab (Humira), brodalumab (Siliq), certolizumab pegol (Cimzia) etanercept (Enbrel), guselkumab (Tremfya), infliximab (Remicade), ixekizumab (Taltz), secukinumab (Cosentyx), tildrakizumab (Ilumya), and ustekinumab (Stelara).
An enzyme inhibitor. The medication apremilast (Otezla) is a new kind of drug for long-term inflammatory diseases like psoriasis and psoriatic arthritis. It's a pill that blocks a specific enzyme, which helps to slow other reactions that lead to inflammation.
PHOTOTHERAPY
Phototherapy or light therapy, involves exposing the skin to ultraviolet light on a regular basis and under medical supervision. Treatments are done in a doctor's office or psoriasis clinic or at home with phototherapy unit. The key to success with light therapy is consistency.
National Psoriasis Foundation does not support the use of indoor tanning beds as a substitute for phototherapy performed with a prescription and under a doctor's supervision. Indoor tanning raises the risk of melanoma by 59 percent, according to the American Academy of Dermatology and the World Health Organization, and does not provide the type of light that most effectively treats psoriasis. Read more on the Psoriasis Foundation position on indoor tanning beds »
Find a provider who offers phototherapy in our Health Care Provider Directory »
Learn more about phototherapy by contacting our Patient Navigation Center »
- Learn about different types of light therapy.
Ultraviolet light B (UVB)
- Sunlight
- Psoralen + UVA (PUVA)
- Laser Treatments
- Tanning beds
ULTRAVIOLET LIGHT B (UVB)
UVB PHOTOTHERAPY
Present in natural sunlight, ultraviolet B (UVB) is an effective treatment for psoriasis. UVB penetrates the skin and slows the growth of affected skin cells. Treatment involves exposing the skin to an artificial UVB light source for a set length of time on a regular schedule. This treatment is administered in a medical setting or at home.
There are two types of UVB treatment, broad band and narrow band. The major difference between them is that narrow band UVB light bulbs release a smaller range of ultraviolet light. Narrow-band UVB is similar to broad-band UVB in many ways. Several studies indicate that narrow-band UVB clears psoriasis faster and produces longer remissions than broad-band UVB. It also may be effective with fewer treatments per week than broad-band UVB.
During UVB treatment, your psoriasis may worsen temporarily before improving. The skin may redden and itch from exposure to the UVB light. To avoid further irritation, the amount of UVB administered may need to be reduced. Occasionally, temporary flares occur with low-level doses of UVB. These reactions tend to resolve with continued treatment.
UVB can be combined with other topical and/or systemic agents to enhance efficacy, but some of these may increase photosensitivity and burning, or shorten remission. Combining UVB with systemic therapies may increase efficacy dramatically and allow for lower doses of the systemic medication to be used.
UVB treatment is offered in different ways. This can include small units for localized areas such as the hands and feet, full body units or handheld units. Some UVB units use traditional UV lamps or bulbs, and others use LED bulbs.
HOME UVB PHOTOTHERAPY
Treating psoriasis with a UVB light unit at home is an economical and convenient choice for many people. Like phototherapy in a clinic, it requires a consistent treatment schedule. Individuals are treated initially at a medical facility and then begin using a light unit at home.
It is critical when doing phototherapy at home to follow a doctor's instructions and continue with regular check-ups. Home phototherapy is a medical treatment that requires monitoring by a health care professional.
All phototherapy treatments, including purchase of equipment for home use, require a prescription. Some insurance companies will cover the cost of home UVB equipment. Vendors of home phototherapy equipment often will assist you in working with your insurance company to purchase a unit.
SUNLIGHT
Although both UVB and ultraviolet light A (UVA) are found in sunlight, UVB works best for psoriasis. UVB from the sun works the same way as UVB in phototherapy treatments.
Short, multiple exposures to sunlight are recommended. Start with five to 10 minutes of noontime sun daily. Gradually increase exposure time by 30 seconds if the skin tolerates it. To get the most from the sun, all affected areas should receive equal and adequate exposure. Remember to wear sunscreen on areas of your skin unaffected by psoriasis.
Avoid overexposure and sunburn. It can take several weeks to see improvement. Have your doctor check you regularly for sun damage.
Some topical medications can increase the risk of sunburn. These include tazarotene, coal tar, Elidel (pimecrolimus) and Protopic (tacrolimus). Individuals using these products should talk with a doctor before going in the sun.
People who are using PUVA or other forms of light therapy should limit or avoid exposure to natural sunlight unless directed by a doctor.
PSORALEN + UVA (PUVA)
Like UVB, ultraviolet light A
(UVA) is present in sunlight. Unlike UVB, UVA is relatively ineffective unless used with a light-sensitizing medication psoralen, which is administered topically or orally. This process, called PUVA, slows down excessive skin cell growth and can clear psoriasis symptoms for varying periods of time. Stable plaque psoriasis, guttate psoriasis, and psoriasis of the palms and soles are most responsive to PUVA treatment.
[caption id="attachment_1758" align="aligncenter" width="550"]
 PUVA
PUVA[/caption]The most common short-term side effects of PUVA are nausea, itching and redness of the skin. Drinking milk or ginger ale, taking ginger supplements or eating while taking oral psoralen may prevent nausea. Antihistamines, baths with colloidal oatmeal products or application of topical products with capsaicin may help relieve itching. Swelling of the legs from standing during PUVA treatment may be relieved by wearing support hose.
LASER TRETMENT
EXCIMER LASER
The excimer laser—recently approved by the Food and Drug Administration (FDA) for treating chronic, localized psoriasis plaques—emits a high-intensity beam of ultraviolet light B (UVB).
The excimer laser can target select areas of the skin affected by mild to moderate psoriasis, and research indicates it is a particularly effective treatment for scalp psoriasis. Researchers at the University of Utah, for example, reported in The Journal of Drugs in Dermatology that in a small series of patients, laser treatment, combined with a topical steroid, cleared scalp psoriasis that resisted other treatment.
Individual response to the treatment varies. It can take an average of four to 10 sessions to see results, depending on the particular case of psoriasis. It is recommended that patients receive two treatments per week, with a minimum of 48 hours between treatments.
There is not yet enough long-term data to indicate how long the improvement will last following a course of laser therapy.
TANNING BEDS
Some people visit tanning salons as an alternative to natural sunlight. Tanning beds in commercial salons emit mostly UVA light, not UVB. The beneficial effect for psoriasis is attributed primarily to UVB light. National Psoriasis Foundation does not support the use of indoor tanning beds as a substitute for phototherapy performed with a prescription and under a doctor's supervision. Read more on the Psoriasis Foundation position on indoor tanning bed.
The American Academy of Dermatology, the Food and Drug Administration (FDA) and the Centers for Disease Control and Prevention all discourage the use of tanning beds and sun lamps. Indoor tanning raises the risk of melanoma by 59 percent, according to the American Academy of Dermatology and the World Health Organization. In May 2014, the FDA reclassified sunlamps (which are used in tanning beds and booths) from Class I (low risk) to Class II (moderate risk) products. The FDA can exert more regulatory control over Class II products, according to a press release on the FDA website.
The ultraviolet radiation from these devices can damage the skin, cause premature aging and increase the risk of skin cancer.



 PUVA[/caption]The most common short-term side effects of PUVA are nausea, itching and redness of the skin. Drinking milk or ginger ale, taking ginger supplements or eating while taking oral psoralen may prevent nausea. Antihistamines, baths with colloidal oatmeal products or application of topical products with capsaicin may help relieve itching. Swelling of the legs from standing during PUVA treatment may be relieved by wearing support hose.
PUVA[/caption]The most common short-term side effects of PUVA are nausea, itching and redness of the skin. Drinking milk or ginger ale, taking ginger supplements or eating while taking oral psoralen may prevent nausea. Antihistamines, baths with colloidal oatmeal products or application of topical products with capsaicin may help relieve itching. Swelling of the legs from standing during PUVA treatment may be relieved by wearing support hose.